
Remember, however, that a hydrogen atom has only a single pair of electrons around it.Ĥ. Complete the octets around all the atoms bonded to the central atom. In other cases, you may need more information before you can draw the Lewis structure.ģ. 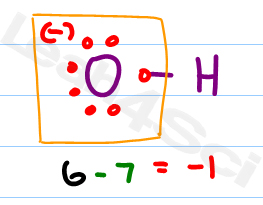
Remember that the central atom is generally less electronegative than the atoms surrounding it. In many polyatomic molecules and ions, the central atom is usually written first, as in and SF 4. The formula HCN, for example, tells you that the carbon atom is bonded to the H and to the N. Chemical formulas are often written in the order in which the atoms are connected in the molecule or ion. Write the symbols for the atoms, show which atoms are attached to which, and connect them with a single bond ( a dash, representing two electrons). Do not worry about keeping track of which electrons come from which atoms. 
For a cation, subtract one electron from the total for each positive charge. (Use the periodic table to help you determine the number of valence electrons in each atom.) For an anion, add one electron to the total for each negative charge. Sum the valence electrons from all atoms. To do so, you should use the following procedure.ġ. For this reason, drawing Lewis structures is an important skill that you should practice. Lewis structures can help us understand the bonding in many compounds and are frequently used when discussing the properties of molecules. CHEMISTRY THE CENTRAL SCIENCE 8 BASIC CONCEPTS OF CHEMICAL BONDING 8.5 DRAWING LEWIS STRUCTURES
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
